
Reversing a Model of Parkinson’s Disease With in Situ Converted Nigral Neurons
Qian H, Kang X, Hu J, Zhang D, Liang Z, Meng F, Zhang X, Xue Y, Maimon R, Dowdy SF, Devaraj NK, Zhou Z, Mobley WC, Cleveland DW, Fu XD.
Joint First Authors

Dr. Hao Qian
Parkinson’s Disease (PD) is a progressive neurodegenerative disease characterised by loss of dopaminergic neurons within the substantia nigra (a region of the midbrain). The main symptoms are tremors and muscle stiffness due to loss of dopamine signalling. Current treatments can ameliorate symptoms to a degree, but no cure is available.
For more information on PD, please see this explanation:
Qian and colleagues from the University of California, San Diego report on their efforts to replace the lost dopaminergic neurons by transdifferentiating astrocytes into functional neurons. From their previous work they knew that in fibroblasts the RNA-binding protein (PTB) inhibits expression of many neuronal genes, including its neuronal analogue nPTB. Neuronal PTB in turn inhibits the transcription factor BRN2 and miR-9 which are required for neuronal maturation. Downregulation of PTB using a lentiviral small hairpin RNA (shRNA) in both mouse and human astrocytes resulted in a transient upregulation of nPTB and subsequent expression of pan-neuronal markers in 50-80% of treated cells. These converted neurons were functional and responded to synaptic inputs from other neurons in co-culture.
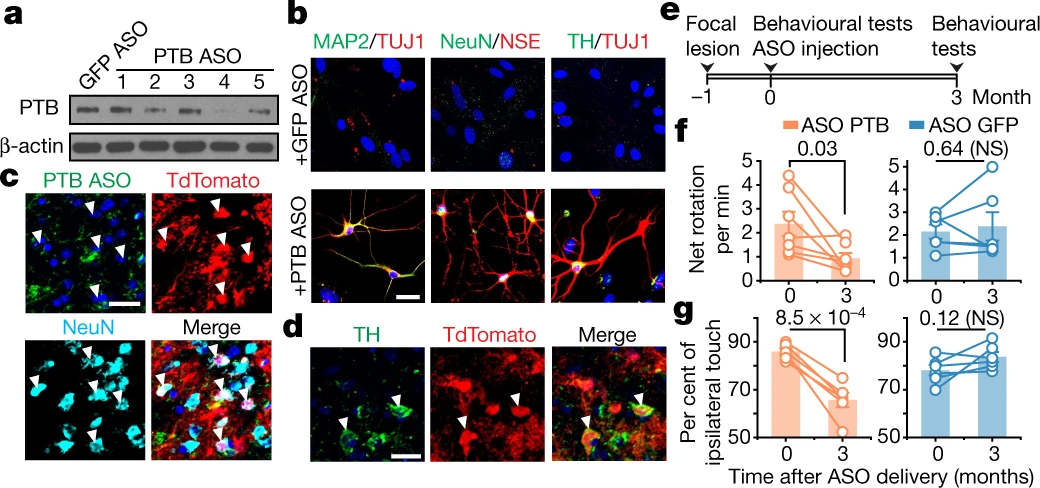
Injection of AAV2-PTB-shRNA into mouse midbrains resulted in ~80% of infected astrocytes expressing neuronal markers 10 weeks after treatment. Around 30% of these converted neurons demonstrated dopaminergic neuron markers and morphology. The neurons were appropriately localized, showed normal functionality, and were successfully integrated into the nigrostriatal pathway over time. Treatment with the AAV-PTB shRNA also restored neuronal function and disease-associated motor phenotypes in the 6-OHDA PD mouse model.
 The authors obtained similar results with ASOs targeting PTB, demonstrating that transient suppression of PTB is sufficient to convert astrocytes into functional dopaminergic neurons. Although many challenges remain before clinical translation, antisense oligonucleotides have big advantages over viral vector mediated shRNA – significantly simplified production and thus vastly reduced cost as well as ease of repeat administration would clearly make them the method of choice.
The authors obtained similar results with ASOs targeting PTB, demonstrating that transient suppression of PTB is sufficient to convert astrocytes into functional dopaminergic neurons. Although many challenges remain before clinical translation, antisense oligonucleotides have big advantages over viral vector mediated shRNA – significantly simplified production and thus vastly reduced cost as well as ease of repeat administration would clearly make them the method of choice.
Why you should read it
Converting astrocytes to neurons using a straightforward ASO treatment is a highly promising approach to cure Parkinson’s Disease and potentially other neurodegenerative diseases.