
August 2020 Joint Papers of the Month
Antisense oligonucleotide modulation of non-productive alternative splicing upregulates gene expression
Lim KH, Han Z, Jeon HY, Kach J, Jing E, Weyn-Vanhentenryck S, Downs M, Corrionero A, Oh R, Scharner J, Venkatesh A, Ji S, Liau G, Ticho B, Nash H, Aznarez I.
Joint First Authors

Dr. Zhou Han

Prof. Stephen J. Thomas
Senior Authors

Dr. Huw Nash

Dr. Barry Ticho

Dr. Isabel Aznarez
Antisense oligonucleotides increase Scn1a expression and reduce seizures and SUDEP incidence in a mouse model of Dravet syndrome.
Han Z, Chen C, Christiansen A, Ji S, Lin Q, Anumonwo C, Liu C, Leiser SC, Meena, Aznarez I, Liau G, Isom LL.
First authors

Dr. Zhou Han

Chunling Chen

Charles Anumonwo

Chante Liu
Senior Authors

Dr. Isabel Aznarez

Prof. Lori L. Isom
Dravet syndrome (OMIM 607208) is a severe developmental and epileptic encephalopathy resulting in developmental delays and intellectual disability. About 20% of patients do not reach adulthood due to sudden unexpected death in epilepsy (SUDEP) or status epilepticus. The majority of Dravet syndrome cases are due to de novo heterozygous mutations in the SCN1A gene, which codes for the α subunit of the voltage-gated sodium channel NaV1.1. Although current therapies can provide partial seizure management, they do not address the underlying SCN1A haploinsufficiency which is directly associated SUDEP.
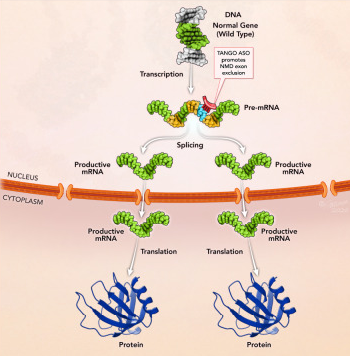
These two papers now report a novel application of antisense oligonucleotides, Targeted Augmentation of Nuclear Gene Output (TANGO). It uses splice-switching oligonucleotides to suppress non-productive alternative splicing events, resulting in proportional upregulation of the correctly spliced isoform and thus increased target protein expression.
For more information on Dravet syndrome and how TANGO works, please watch this presentation by Dr. Barry Ticho:
Lim and colleagues from Stoke Therapeutics firmly establish the wide applicability of TANGO across different alternative splicing events, target genes, ASO chemistries (PMO and 2′MOE-PS) and cell types as well as in a mouse model. They demonstrate that TANGO reduced the mRNA isoform destined for nonsense mediated decay and increased the productive mRNA isoform proportionally. This change in isoform ratios resulted in increased functional protein expression in all cases.
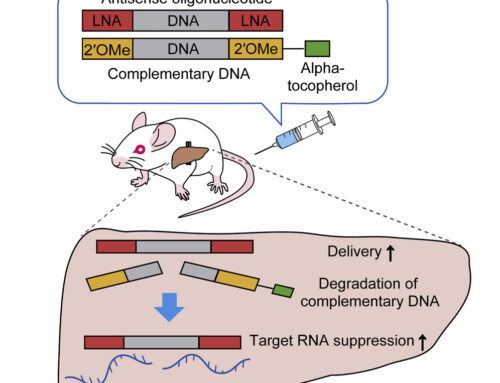
Han et al. then show that TANGO has therapeutic effects in a mouse model of Dravet syndrome. A single intracerebroventricular injection of the therapeutic ASO at postnatal day 2 led to a dose-dependent increase in productive Scn1a transcripts and NaV1.1 protein. This increase was still present 30 days after treatment. Treatment resulted in significantly increased survival (97% versus 23% in the control group) at 90 days as well as significantly fewer seizures. Latency to seizures was prolonged in the treatment group.
Together, these two papers clearly demonstrate the Tango’s therapeutic value in upregulating protein expression without exceeding endogenous levels.
Why you should read it
This new therapeutic use of antisense oligonucleotides will be widely applicable to diseases that are caused mutations leading to loss of protein function.
Please also check out short activating RNAs for oligonucleotide therapeutics that can upregulate protein expression.