Reversal of siRNA-mediated gene silencing in vivo
Zlatev I, Castoreno A, Brown CR, Qin J, Waldron S, Schlegel MK, Degaonkar R, Shulga-Morskaya S, Xu H, Gupta S, Matsuda S, Akinc A, Rajeev KG, Manoharan M, Maier MA, Jadhav V.
Nat Biotechnol. 2018 Jul;36(6):509-511.
Clinical data from GalNAc-conjugated siRNAs demonstrate long-term persistence of their therapeutic effect (up to 6 months after a single injection). Although such durability is truly impressive and a positive attribute, the option to reverse or exert finer control over the pharmacology would be desirable.
See this previous OTS Perspective for more detail on pharmacology of GalNAc-conjugated siRNA inclisiran or watch an explanation of how RNA interference (RNAi) works:
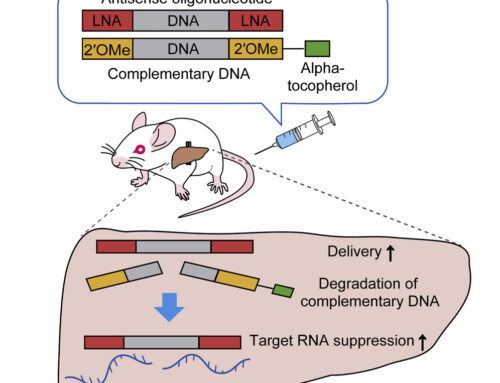
Here, Zlatev et al. from Alnylam Pharmaceuticals report on the development of REVERSIRs, short high-affinity oligonucleotides complementary to the siRNA guide strand which mediate rapid reversal of RNAi activity. In a series of experiments, the authors elucidate guidelines for the design of these REVERSIRs. All REVERSIRs were linked to the same trivalent GalNAc ligand as GalNAc–siRNAs via a nuclease-cleavable phosphodiester linkage at the 3′ end. REVERSIRs have a fully phosphorothioate (PS)-modified backbone as well as 2′-O-methyl and locked nucleic acid (LNA) modifications.
As expected, increasing the LNA content raised the melting temperature (hybridization affinity) of REVERSIRs annealed with siRNA guide strands and improved reversal of siRNA-mediated target RNA knockdown in vitro and in vivo. REVERSIRs containing five LNA modifications mediated rapid and full reversal of siRNA activity four days after a 0.1 mg/kg REVERSIR dose in mice previously treated with 3 mg/kg TTR–siRNA. An LNA modification at the nucleotide complementary to position 6 of the target guide strand proved to be of particular importance. Shorter REVERSIRs (8- and 9-mers) showed the strongest reversal of siRNA activity. Full coverage of the entire guide seed region with the REVERSIR sequence, GalNAc-assisted targeted delivery and the intracellular nuclease-cleavable linker between ligand and oligonucleotide were required for optimal RNAi reversal. Potential REVERSIR off-target binding caused no significant changes in global RNA expression.
In a companion paper, REVERSIRs were used to demonstrate that hepatotoxicity caused by some siRNAs can largely be attributed to RNAi-mediated hybridization based off-target effects with little or no contribution from chemical modifications or the perturbation of RNAi pathways.
Why you should read it
REVERSIRs enable better control of siRNA pharmacology and results should be applicable to other oligonucleotide therapeutics, such as anti-miRs.