
Phase 1 Trial of an RNA Interference Therapy for Acute Intermittent Porphyria
Sardh E, Harper P, Balwani M, Stein P, Rees D, Bissell DM, Desnick R, Parker C, Phillips J, Bonkovsky HL, Vassiliou D, Penz C, Chan-Daniels A, He Q, Querbes W, Fitzgerald K, Kim JB, Garg P, Vaishnaw A, Simon AR, Anderson KE.
N Engl J Med. 2019 Feb 7;380(6):549-558.
The three lead authors:

Eliane Sardh

Manisha Balwani

Pauline Harper
Acute intermittent porphyria (AIP) is caused by mutations in hydroxymethylbilane synthase (HBMS), an enzyme critical in heme biosynthesis. Reduced hepatic heme leads to up-regulation of delta aminolevulinate synthase 1 (ALAS1) expression and subsequent accumulation of neurotoxic delta aminolevulinic acid (ALA) and porphobilinogen (PBG). Although prevalence is high (1:1700), penetrance is low (10%) and only 5-8% of symptomatic patients suffer from repeated attacks characterised by severe abdominal pain, tachycardia, seizures or paresis.
See below or click here for more information (from 1 h).
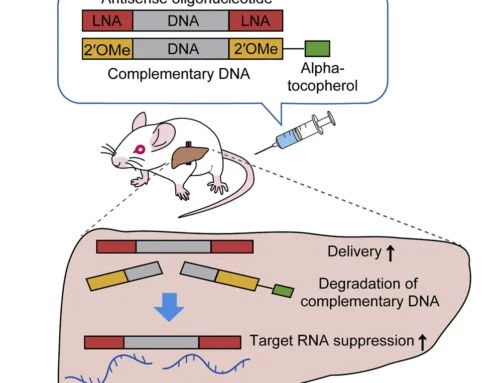
Sardh, Harper and Balwani et al. provide the first clinical proof of concept data for the siRNA therapeutic givosiran (Alnylam) which targets the ALAS1 mRNA.
In this study, patients without attacks in the 6 months before baseline received a single (0.035-2.5 mg/kg) or two doses (0.35 or 1.0 mg/kg, 28 days apart) of givosiran. In a third double-blind part, patients with recurrent attacks (≥2 attacks within 6 months) received 2.5 or 5 mg/kg monthly or quarterly for a total of four and two doses. The treatment was generally well tolerated with no discontinuations during the trial.
A single 2.5 mg/kg givosiran dose resulted in rapid, dose-dependent reductions in urinary ALAS1 mRNA level (86%), urinary ALA (91%) and PBG levels (96%). Repeat doses of 1 mg/kg 28 days apart caused similar decreases, with levels remaining below baseline at day 70. In patients with recurrent attacks, four monthly doses of 2.5 or 5 mg/kg resulted in ALAS1 mRNA reductions of 67% and 74%, respectively. ALA and PBG levels were reduced>90% from baseline. Annualized attack rate among patients who received givosiran was 7.2, compared to 16.7 in the placebo group and annualized number of hemin doses was 12.1 in the givosiran versus 23.4 in the placebo group. There was a clear association between lower ALA levels and reduced annualized attack rate.
Why you should read it
This is the first clinical proof of concept of givosiran demonstrating impressive therapeutic efficacy; results from the follow-on phase III trial have become available this month.