Page 1 of 212
Coronavirus Highlights Urgent Need for Science-Backed Research
News Release from Mary Ann Liebert, Inc. publishers Coronavirus Highlights Urgent Need for Science-Backed Research As an academic publisher, we publish many important resources relevant to COVID-19 such as Health Security, Viral Immunology, Telemedicine and e-Health, and Vector Borne and […]

March 20, 2020
Base-pair Conformational Switch Modulates miR-34a Targeting of Sirt1 mRNA
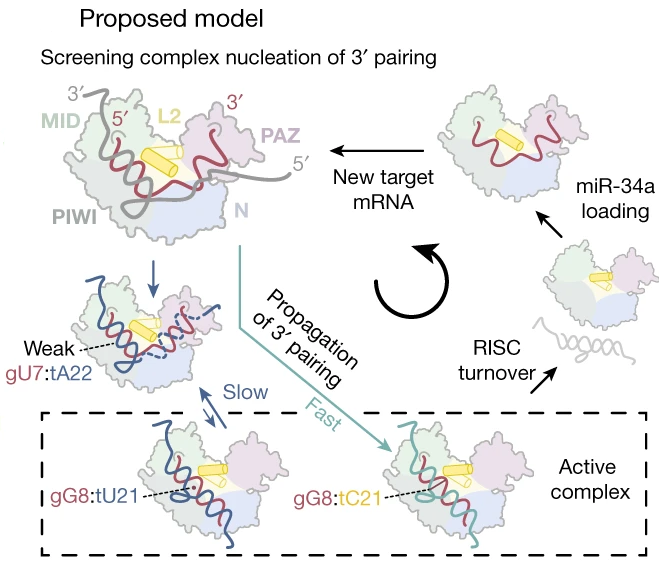
Baronti L, Guzzetti I, Ebrahimi P, Friebe Sandoz S, Steiner E, Schlagnitweit J, Fromm B, Silva L, Fontana C, Chen AA, Petzold K. Nature. 2020 May 27 The first author: As part of RNA-induced silencing complexes (RISCs), microRNAs bind to […]
June 15, 2020
mRNA-encoded, constitutively active STINGV155M is a potent genetic adjuvant of antigen-specific CD8+ T cell response
Papers of interest to society members in the field of oligonucleotide therapeutics. Antisense, aptamers, lncRNA, siRNA, splice modulation and ribozymes.
April 23, 2021
Systematic screening identifies therapeutic antisense oligonucleotides for Hutchinson-Gilford progeria syndrome
Papers of interest to society members in the field of oligonucleotide therapeutics. Antisense, aptamers, lncRNA, siRNA, splice modulation and ribozymes.
Page 1 of 212
Page 1 of 212
Coronavirus Highlights Urgent Need for Science-Backed Research
News Release from Mary Ann Liebert, Inc. publishers Coronavirus Highlights Urgent Need for Science-Backed Research As an academic publisher, we publish many important resources relevant to COVID-19 such as Health Security, Viral Immunology, Telemedicine and e-Health, and Vector Borne and […]

March 20, 2020
Base-pair Conformational Switch Modulates miR-34a Targeting of Sirt1 mRNA
Baronti L, Guzzetti I, Ebrahimi P, Friebe Sandoz S, Steiner E, Schlagnitweit J, Fromm B, Silva L, Fontana C, Chen AA, Petzold K. Nature. 2020 May 27 The first author: As part of RNA-induced silencing complexes (RISCs), microRNAs bind to […]

June 15, 2020
mRNA-encoded, constitutively active STINGV155M is a potent genetic adjuvant of antigen-specific CD8+ T cell response
Papers of interest to society members in the field of oligonucleotide therapeutics. Antisense, aptamers, lncRNA, siRNA, splice modulation and ribozymes.
April 23, 2021
Systematic screening identifies therapeutic antisense oligonucleotides for Hutchinson-Gilford progeria syndrome
Papers of interest to society members in the field of oligonucleotide therapeutics. Antisense, aptamers, lncRNA, siRNA, splice modulation and ribozymes.
Page 1 of 212


